| Program | Indication | Preclinical | Phase 1 | Phase 2 | Phase 2b/3 |
|---|---|---|---|---|---|
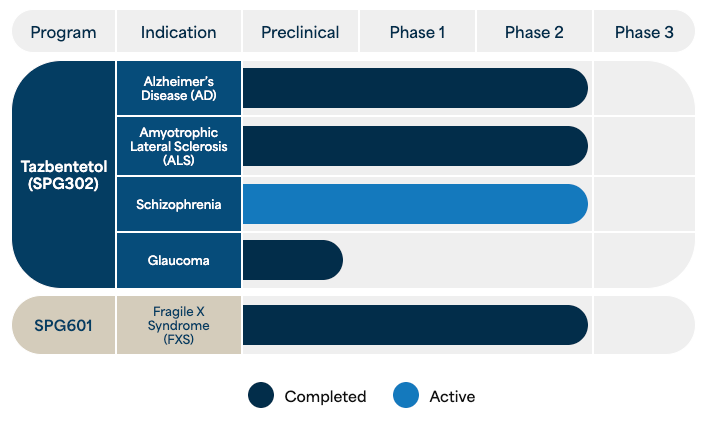
| Tazbentetol (SPG302) |
Alzheimer’s Disease (AD) |
||||
| Amyotrophic Lateral Sclerosis (ALS) |
|||||
| Schizophrenia (SCZ) |
|||||
| Glaucoma | |||||
| Undisclosed | |||||
| SPG601 | Fragile X Syndrome (FXS) |
||||
Completed
Active
Tazbentetol (SPG302)
- Alzheimer’s Disease (AD) (NCT06427668, NCT06833281) – the most common form of dementia worldwide
- Phase 2a completed with topline data presented at Alzheimer’s Association International Conference (AAIC), July 2025, and international Clinical Trials on Alzheimer’s Disease (CTAD), Dec. 2025
- Amyotrophic Lateral Sclerosis (ALS) (NCT05882695, NCT06903286) – the most common adult-onset motor neuron disease
- Phase 2a completed with topline data presented at Northeast ALS Consortium (NEALS), Oct. 2025 and international Motor Neurone Disease Association (MNDA), Dec. 2025
- FDA & EMA Orphan designation in ALS
- Schizophrenia (SCZ) (NCT06442462) – the most prevalent psychotic disorder worldwide
- Phase 2 trial completed enrollment. Interim results will be presented at Schizophrenia International Research Society (SIRS), Mar. 2026
- Glaucoma (Pre-Clinical)
- Undisclosed (Pre-Clinical)
SPG601
- Fragile X Syndrome (FXS) (NCT06413537) – the most common cause of intellectual disability and autism
- Phase 2a (single dose cross-over design) completed, results presented at American Academy of Child and Adolescent Psychiatry (AACAP), 2025, American College of Neuropsychopharmacology (ACNP), 2026
- FDA & EMA Orphan designation in FXS
- FDA Fast Track Designation